 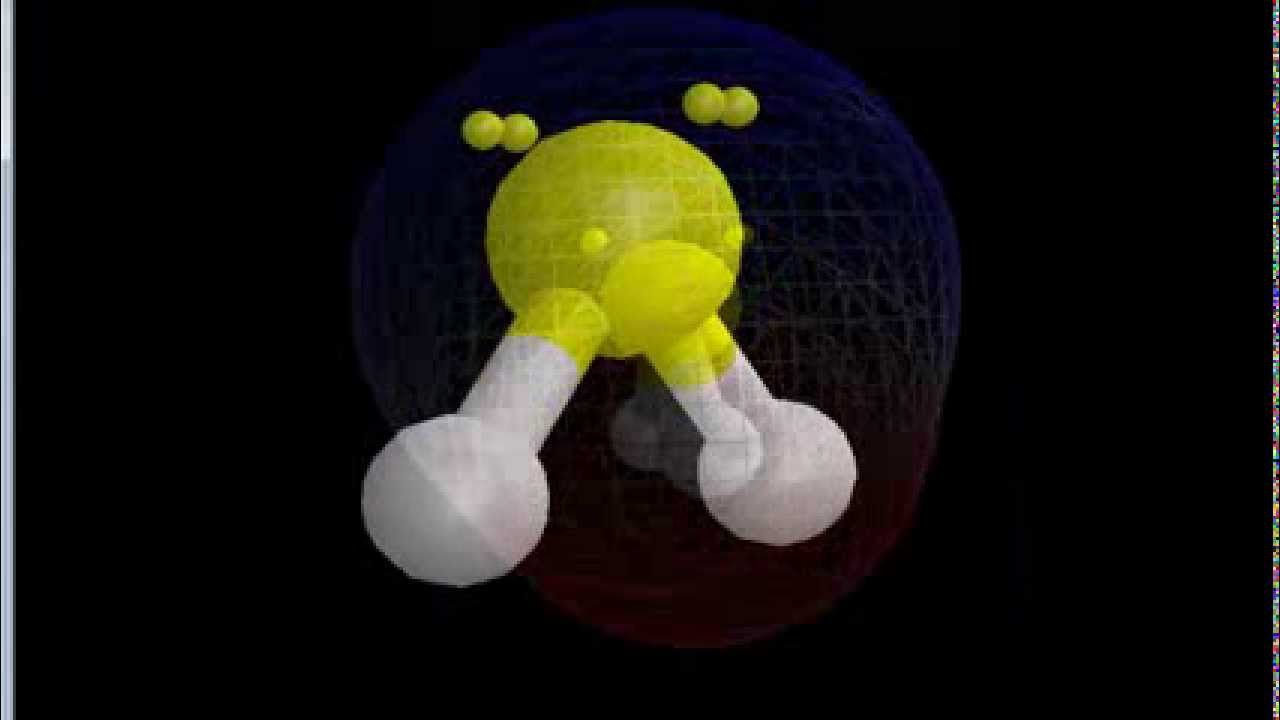
Examples of covalent compounds are H 2O (water), CH 4 (methane) and C 6H 12O 6 (glucose).ĭetermine for each of the following compounds whether they are covalent compounds or not. In the next sections we will look at how to “balance” covalent compounds and how to give them a name.Ĭovalent compounds are those compounds that are entirely made out of non-metal atoms (read the section on metals and non-metals). We will leave the realm of ionic compounds behind and we will focus on covalent compounds.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
